Please provide your technical specifications and project requirements for the Hermetically Sealed Automatic Door. Our engineers will review your application and provide a detailed manufacturing quote, including exact pricing and lead times.
Hermetically Sealed Automatic Door
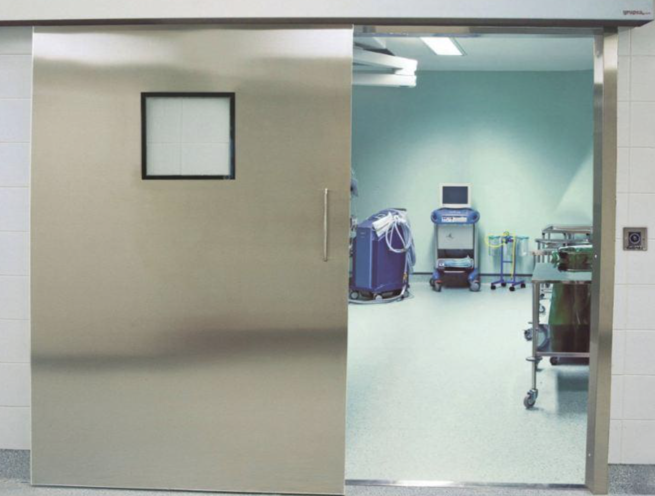
The Madoors USA Automatic Motorized Hermetic Sliding Door is a specially designed airtight sealing sliding door system for pharmaceutical facilities, cleanrooms, operating theatres, food preparation facilities, and laboratories — providing over 99% sealing effectiveness confirmed by official independent testing to reduce cross-contamination, prevent dirty air from entering the cleanroom, and reduce expensive air handling costs. Powered by Madoors USA electronic card and PLC operator, providing hermetic closure in both positive pressure and negative pressure conditions for simultaneous quarantine door and operating room door capability, with non-touch automated passage, floor guiders outside the clear opening without a sill, and full hygiene-cleanable construction, the Madoors USA Hermetic Sliding Door is the definitive infection control entrance solution for the most demanding clinical and pharmaceutical environments.
TECHNICAL SPECIFICATIONS
The Madoors USA Hermetic Sliding Door achieves over 99% sealing effectiveness — independently tested and officially certified — providing the airtight separation between contamination zones required for pharmaceutical cleanroom GMP compliance, hospital operating theatre infection control, and laboratory containment integrity. The 99%+ sealing performance is the primary specification criterion for hermetic doors in environments where the consequence of seal failure is product contamination, infection transmission, or containment breach.
The door operates in both pressure directions — the seal maintains integrity under both positive pressure — where the protected room is pressurized above the surrounding area, as in pharmaceutical cleanrooms and isolation rooms for infection containment — and negative pressure — where the protected room is maintained below the surrounding atmospheric pressure, as in isolation rooms for infectious disease patients and fume laboratories. This bidirectional pressure sealing allows the same door to serve both positive-pressure clean manufacturing environments and negative-pressure isolation and containment environments without modification.
The PLC operator and electronic card system provide the automation integration required for cleanroom and hospital environments — fully automated non-touch door passage eliminates the contamination risk of hand contact with door hardware. The automation is fully integrated into the door concept — the sensor, operator, and seal system function as a unified design rather than a commercial sliding door with added automation.
The door blade weight provides the downward force on the floor seal that ensures firm, solid sealing at the floor contact surface — the weight of the door panel itself contributes to the sealing force without requiring separate mechanical compression systems. This weight-assisted floor sealing directly contributes to the 99%+ seal performance.
Floor guiders outside the clear opening — without a floor sill within the opening — provide the unobstructed floor transition required for bed, trolley, and wheelchair passage without the trip hazard and wheel obstacle that threshold-mounted guide systems create. This sill-free floor guidance is the critical access design requirement for operating theatre and hospital ward doors where beds and medical equipment must pass without obstruction.
KEY FEATURES
Over 99% Sealing Effectiveness — Officially Tested & Certified
The 99%+ sealing effectiveness confirmed by official independent testing is the primary performance specification that differentiates the Madoors USA hermetic door from standard sliding doors marketed as hygienic — the tested and certified sealing performance provides the regulatory compliance documentation required for GMP pharmaceutical facility qualification, hospital operating theatre certification, and laboratory containment validation. The official test result directly demonstrates that the door achieves the specified contamination control performance rather than relying on manufacturer claims.
Bidirectional Pressure Sealing — Quarantine & Operating Room Combined
The airtight sealing performance under both positive overpressure and negative underpressure allows the same door to serve both cleanroom isolation and patient isolation applications — the operating theatre requires positive pressure to prevent contaminated external air from entering the sterile field, while the infectious disease isolation room requires negative pressure to prevent contaminated internal air from escaping to the corridor. A door that seals only in one pressure direction cannot serve both functions — the Madoors USA hermetic door satisfies both criteria simultaneously.
Non-Touch Automated Passage — Infection Control Compliance
The fully automated non-touch door passage eliminates every hand contact point from the door operation — no door handle, push plate, or opening button that contaminated hands would touch. This non-touch operation directly supports the infection control objective of the hermetic door installation — cross-contamination at the door position is eliminated both by the airtight seal when closed and by the contact-free operation during passage.
Sill-Free Floor Guiders — Bed, Trolley & Wheelchair Access
The floor guiders positioned outside the clear door opening — without a sill or threshold within the passage width — provide completely unobstructed floor passage for hospital beds, medical equipment trolleys, pharmaceutical production carts, and wheelchairs. The absence of any floor obstacle at the passage width is the critical accessibility and operational efficiency specification for operating theatre, ICU, and pharmaceutical cleanroom doors where large equipment must pass through the door opening efficiently and safely.
Hygienic Cleanable Construction — Infection Control Maintenance
The hygienic cleanable construction of all door surfaces, frame profiles, and operator components allows thorough cleaning and disinfection with standard healthcare and pharmaceutical cleaning agents — without the concealed joints, porous surfaces, or construction gaps that harbor microbial contamination and resist cleaning. This cleanable design directly supports the routine cleaning protocols required to maintain contamination control at the door position between seal integrity periods.
Simple Rail Construction — 100% Hermetic Closure Reliability
The simple and effective rail construction that provides 100% hermetic closure reliability through the door blade weight sealing mechanism eliminates the mechanical complexity of alternative compression seal systems — fewer mechanical components means fewer failure modes, simpler maintenance, and more reliable long-term seal performance. The rail construction reliability directly supports the continuous contamination control requirement of pharmaceutical and clinical installations where door seal failure has immediate product quality or patient safety consequences.
Multiple Opening Positions — Nurse, Trolley & Bed
The choice of door opening positions — nurse access width for individual personnel passage, trolley width for medication and equipment carts, and bed width for patient bed transfer — allows the automation to be programmed for the specific passage requirements of each clinical zone without requiring full door travel for every access event. This position selection directly reduces the air exchange between zones at each passage event — smaller opening positions admit less uncontrolled air than full door travel for every passage.
Full Automation Integration — PLC & Electronic Card Control
The full integration of automation into the door concept — PLC controller, electronic card access, sensor detection, and seal mechanism all designed as a unified system — provides the reliability and performance consistency required for pharmaceutical and clinical environments. Automation that is integrated into the door design from the beginning provides more reliable performance than automation added to a standard commercial door, directly supporting the continuous availability requirements of 24-hour clinical and pharmaceutical operations.
APPLICATION ENVIRONMENTS
Operating Theatres — Positive pressure hermetic sealing preventing contaminated corridor air from entering the sterile surgical field. Non-touch automated passage for gowned surgical teams. Bed width opening for patient transfer. Bidirectional pressure capability for combined clean and isolation use.
Pharmaceutical Cleanrooms — Positive pressure hermetic sealing for GMP Grade A, B, C, and D cleanroom compliance. Airtight particle control preventing cleanroom contamination from less-controlled adjacent areas. PLC integration with cleanroom management systems.
Hospital Isolation Rooms — Negative pressure hermetic sealing containing infectious patient room air within the isolation environment. Bidirectional pressure capability for flexible positive/negative pressure zone management.
Laboratories & Research Facilities — Containment integrity for BSL-2, BSL-3, and chemical laboratory environments. Non-touch automated passage for PPE-wearing laboratory personnel. Sill-free access for laboratory equipment trolleys.
Food Processing Facilities — Airtight sealing between processing zones of different hygiene classification. Cleanable construction for food contact environment cleaning and sanitation protocols. Non-touch passage eliminating contamination at zone boundaries.
ASSEMBLY & INSTALLATION SEQUENCE
Environment Assessment & Specification
Room pressure classification — positive or negative — required sealing performance level, passage width requirements — nurse, trolley, or bed — access control integration, cleaning protocol compatibility, and PLC interface requirements are confirmed.
Frame Installation
The hermetic door frame is installed in the structural opening — frame-to-wall seal continuity at all frame perimeter positions is verified. Floor guider installation outside the clear opening is completed.
Door Blade & Rail Installation
The door blade is installed on the rail system. Door blade weight contact with the floor seal surface and perimeter seal contact with the frame at all door positions are verified.
PLC, Sensor & Access Control Installation
PLC operator, proximity sensors for non-touch activation, electronic card access control, and opening position programming are installed and configured. Bidirectional pressure seal performance under both positive and negative pressure conditions is tested.
Seal Performance Verification
Independent seal performance testing to confirm 99%+ effectiveness is conducted. Test documentation is compiled for regulatory submission in pharmaceutical qualification and hospital certification processes.
Certification Documentation & Handover
Official seal test certificate, PLC commissioning documentation, GMP compliance documentation, and installation records are compiled into the project handover package.
DEPLOYMENT SCENARIOS & USE CASES
Madoors USA Hermetically Sealed Automatic Doors are specified for any cleanroom, clinical, or pharmaceutical facility requiring certified airtight automated door systems.
- Hospital operating theatre hermetic sliding doors
- Intensive care unit and high-dependency ward isolation doors
- Infectious disease isolation room negative pressure doors
- Pharmaceutical cleanroom GMP Grade A/B/C/D doors
- Sterile manufacturing facility hermetic access doors
- Food processing cleanroom zone separation doors
- Microelectronics cleanroom contamination control doors
- Medical device manufacturing cleanroom access doors
- Hospital pharmacy sterile preparation room doors
- Research laboratory BSL-2 and BSL-3 containment doors
- Radiopharmacy hot laboratory hermetic access doors
- Vaccine production facility cleanroom entrance doors
- Hospital central sterile supply department doors
- Biotech and genomics laboratory contamination control doors
- Any cleanroom or clinical environment requiring 99%+ certified hermetic airtight automated sliding door
- Category: Automatic Door